Click Here to See Update 3/26/2020
In scaling up the medical infrastructure to handle the surge in COVID-19 patients, one of the critical capacity constraints is the number of ventilators available. To date, the focus has been on acquiring more ventilator devices. While this does make sense, it will take time to manufacture such devices.
Let’s look at this problem slightly differently. Instead of thinking of the problem as, “How do we increase the number of ventilators,” it makes more sense to define the problem as, “How do we increase the number of patients who can be ventilated?”
Let me explain.
Under normal protocols, these two numbers are the same. One ventilator device is dedicated to one patient. Therefore, the number of ventilators and the number of patients being ventilated are one and the same.
Here’s the key insight I’ve recently come across.
Most full-featured ventilators are set to 25%-35% of the maximum level of air they can move per breath. Most of the device’s ventilation capacity goes unused under normal usage protocols.
In fact, most full-featured ventilators have the underlying ability to ventilate up to four patients simultaneously — but they aren’t configured to do so by default.
Full-featured ventilators can provide 2,000 milliliters of tidal volume. Tidal volume is the volume of air you can breathe in and then out in a single breathing cycle. 2,000 ml is approximately the same volume as a half-gallon-sized carton of milk.
When you inhale, do you inhale a half-gallon worth of air?
No. Neither do I. Actually, neither do 95% of patients.
So, how much air do you need to breathe?
Well, it turns out that it depends on your weight.
Heavier patients consume more air than smaller patients. That makes intuitive sense. A 250-lb, 50-year-old male doesn’t consume the same air as a 1‑year-old weighing 20 lbs.
There’s a formula for this. For an otherwise healthy patient in need of ventilation, they should receive no more than 10 milliliters of air from a ventilator per 1 kilogram of body weight.
For a patient with lung impairment (such as those with pneumonia, which COVID-19 can sometimes cause), too much air per breathing cycle (or tidal volume) can damage the lungs. These patients should receive 6 – 8 ml per kg of ideal body weight per breathing cycle. To simplify the math, let’s call it 7 ml per 1 kilogram of weight.
So, let’s do some math to figure out how much air a ventilator can move.
2,000 ml tidal volume / 7 ml per kg = 285 kg combined patient ideal body weight
285 kg x 2.2 lb per kg = 627 lbs combined patient ideal body weight
In other words, a single ventilator can move enough air to support 627 lbs worth of patient with lung impairment.
For reference, the average adult male in the United States weighs 200 lbs. The average adult female weighs 170 lbs.
This means a single full-featured ventilator has the ability to move enough air to support three average adult males or 3.7 females.
So, the constraint here is not the ventilator machine itself, as most ventilators run at only 25%-35% utilization. It is the ability to share a ventilator amongst multiple patients that is the constraint that is hit first.
By allocating one ventilator to one average-size adult male patient, over 65% of the machine’s capacity to ventilate is not being used. From a scaling capacity standpoint, this is inefficient.
Now, it turns out that the ability to use a “splitter” to route the air pumping capacity of a ventilator to multiple patients has been studied in lung simulators, animal studies, and used in the field — all successfully.
In 2006, Dr. Greg Neyman and Dr. Charlene Babcock, emergency medicine physicians in Detroit, Michigan, did a study on exactly this. They wondered if, during a disaster-related surge of patients, a ventilator could be reconfigured to support multiple patients. Would it work?
Short answer: Yes.
They used standard equipment found in an emergency room to create a two-way and four-way splitter. They then used the ventilator to ventilate four simulated-lung devices for 12 hours. These devices had several sensors to track the output of the ventilator for each “lung.” The data collected showed that the pilot project was successful.
This is the paper they wrote on this technique: A Single Ventilator for Multiple Simulated Patients to Meet Disaster Surge.
This is a video of Dr. Charlene Babcock demonstrating the technique:
Around the same time, Dr. Lorenzo Paladino et al. devised a similar experiment to answer the same question. Instead of using a ventilator to ventilate four lung-simulator devices, they tried out the technique on four human-sized sheep.
They treated the sheep as if they were human patients with lung impairments and breathing problems. The “patients” were sedated, intubated, and hooked up to a ventilator that did the breathing for them.
In short, it worked. The ventilators kept all four “patients” alive for 12 hours. Sedation was turned off and all four patients started to breathe normally again on their own.
This is the paper Dr. Paladino and his colleagues wrote describing their findings: Increasing Ventilator Surge Capacity in Disasters.
For many years, nobody had attempted (or at least hadn’t documented their attempts) to use this approach on human patients.
However, that changed on October 1, 2017. On that day, Dr. Kevin Menes was running the Emergency Department at Sunrise Hospital near Las Vegas, Nevada. In the middle of his shift, the deadliest mass-casualty gun shooting in modern American history took place. In the hours that followed, Dr. Menes and his team (and auxiliary staff) would end up treating over 215 penetrating gunshot wounds… in seven hours. (If this isn’t the definition of a hospital overwhelmed, I don’t know what is.)
As you might imagine, major blood loss impaired many patients’ abilities to breathe and get enough oxygen circulating with whatever amount of blood remained in their bodies.
As the surge of patients flooded the Emergency Room, the hospital had run out of ventilator machines. (Sound familiar?)
In his own words, here’s what Dr. Menes did…
“…the respiratory therapist, said, ‘Menes, we don’t have any more ventilators.’ I said, ‘It’s fine,’ and requested some Y tubing. Dr. Greg Neyman, a resident a year ahead of me in residency, had done a study on the use of ventilators in a mass-casualty situation. What he came up with was that if you have two people who are roughly the same size and tidal volume, you can just double the tidal volume and stick them on Y tubing on one ventilator.”
You see, Dr. Menes had trained in the same hospital as Dr. Neyman and was familiar with his experiment using a splitter on a ventilator.
You can read Dr. Menes’ personal account of that night here: How One Las Vegas ED Saved Hundreds of Lives After the Worst Mass Shooting in U.S. History.
Now, there are several issues with using splitters with ventilator machines.
1. Using a splitter is a non-standard, “off-label” use of the device. It’s not a normal standard of care. Hence, all the studies have suggested such a configuration would only be used in a mass-casualty situation (which is defined as when you have more patients than you can handle). If we’re not at a mass-casualty situation yet, we are certainly on our way there — especially in certain regional COVID-19 hot spots in the United States.
Dr. Babcock proposes the following approach to navigating this issue. To paraphrase, if I have one ventilator and four patients, here’s what I would say to the four families, “Your loved one needs this machine or he/she will likely die. I can pick who gets it, allowing three of your loved ones to die, or you can share the machine. What do you want to do?”
(I suspect that, when faced with the choice of a 75% chance of certain death versus sharing a ventilator, most people would be fine with sharing.)
2. Sharing a single ventilator does create a risk for cross-contamination. You would not want to share a single ventilator between three COVID-19 patients and one non-COVID-19 patient. Such usage would very likely cause the non-COVID-19 patient to be infected. However, if all four patents are already infected, then this risk is mitigated.
3. Grouping patients requires similar tidal volume needs. All two or four patients sharing the ventilator must have similar ventilation needs. When one ventilator is assigned to one patient, the ventilator settings can be adjusted to meet the single patient’s needs very precisely. When two or four patients share a ventilator, you use a single setting for all patients. This means you want patients of a similar body weight (as this determines the volume of air they need per breath). You also want patients with similar degrees of lung impairment as the breathing capacity can vary significantly based on this trait.
4. Patients need to be placed in close proximity to the ventilator, but ICU rooms aren’t designed for that. I’ve only been to the intensive care wards at two hospitals — one community hospital and one research hospital (UC San Francisco). These wards are designed to prevent infection between patients. As a result, each patient has their own room or at least their own alcove (a room with three walls, with the fourth wall “open” and visible to the nurse’s monitoring station).
In other words, you can solve the ventilation problem, but now you have a “real estate” configuration problem to solve.
Let me discuss this fourth issue in more detail. One of the principles of scaling up operations to meet surges in demand is to remove the point of greatest constraint in your system. In the case of COVID-19, ventilation capacity is one of the top constraints. Using the splitter solution, ventilation patient capacity can be increased between two to four times without any change in the number of ventilation units.
However, every time you fix the greatest constraint, you will suddenly discover the second greatest constraint. In the case of scaling patient ventilation capacity, the new constraint becomes “real estate,” or floor space.
Standard intensive care unit configurations (at least the ones I’ve been to) aren’t set up to group two to four really sick patients together. The premise has always been to separate them. While solving one problem (patient ventilation capacity), you create another problem (suboptimal floor space configuration).
However, you’re ahead of the game if you trade-in a major headache for a lesser headache.
In this case, the number of ventilators in the country is more or less fixed in the short-term. However, we have more “real estate” available to us than ventilators. So all else being equal, I’d rather have a solvable “real estate” problem than a ventilator shortage problem. That said, someone still has to go figure out how to solve the “real estate” problem.
5. Legal Liability — It would be extremely helpful if this approach could be validated further in the field and receive an expedited approval, or at least some kind of crisis waiver from the FDA, to use ventilators in this particular way. Until then, it would make sense for hospital general counsels to start drafting “Consent to Treat” forms for this non-standard ventilation usage — just in case things get so bad that doctors and nurses need to use it. This kind of scenario is entirely anticipatable and, when running fast-moving operations of any kind, you want to solve the anticipatable problems early so that your top talent only has to deal with the unanticipated problems.
6. Developing a Three-Way Splitter — All the academic studies and field experiences have either split the air from a ventilator to two or four patients. This is because standard equipment available in an emergency room allows for these configurations. However, since a single full-featured ventilator can ventilate patients with a combined weight of 627 lbs, we could get more efficient capacity utilization with a three-way splitter. For example, a three-way splitter could allocate 627 lbs worth of patient ventilation capacity to three patients weighing approximately 209 lbs or less. The average adult male is 200 lbs. This would serve even more patients than ventilators that only use a two-way or four-way splitter.
7. Scalable Knowledge Transfer — Someone from the National Institutes of Health, Centers for Disease Control and Prevention, Department of Health and Human Services, and state-level health boards should each be contacting Drs. Neyman, Babcock, Paladino, and/or Menes to more precisely document their collective knowledge, write down what they recommend regarding this protocol in specific detail, and disseminate such knowledge to all the hospitals in the country.
Hopefully, things don’t get so bad that we need to do this. But, hope is not a plan. We should prepare by forwarding knowledge of these techniques to those who need to know. This is an essential part of increasing critical-care capacity (a.k.a. “raising the ceiling”) in our country.
COVID-19 moves fast. We need to move faster. While publishing in peer-reviewed journals is the gold standard for introducing protocol innovation in the healthcare field, we do not have the time. In a crisis, perfection is the enemy of good enough.
My goal in writing this article is to bring attention to the findings of Drs. Neyman, Babcock, and Paladino that have since been proven in the field by Dr. Menes. My hope is that others can build on and improve upon their work and have back-up options if we do run out of ventilators. I would appreciate it if you would pass along this message to as many people as possible — especially those who are experts in the medical, intensive care, emergency care, or government health industries.
Update 3/18/2020 9:20 am PT
8. Optimal for One vs. Suboptimal for All — One issue is that a ventilator’s settings are usually tailored to a specific patient’s vital signs. What happens if one patient needs a higher setting that would cause another patient to receive “too much”? In particular, there’s a concern around PEEP (positive end-expiratory pressure). This is the amount of air pressure the ventilator produces at the end of an exhale to prevent the alveoli (oxygen exchange area of the lungs) from “collapsing.” As I understand, this pressure setting varies from patient to patient and isn’t correlated with body weight but more so patient illness status.
The consensus feedback is that this approach clearly isn’t ideal (which is why it is not standard practice). There are no studies on long-term outcomes. However, if desperate enough, such configuration could work as i) a temporary solution until each patient gets their own ventilator, or ii) becomes a difficult triage decision: Is it better to treat four patients suboptimally or one patient optimally?
Examples and Field Reports
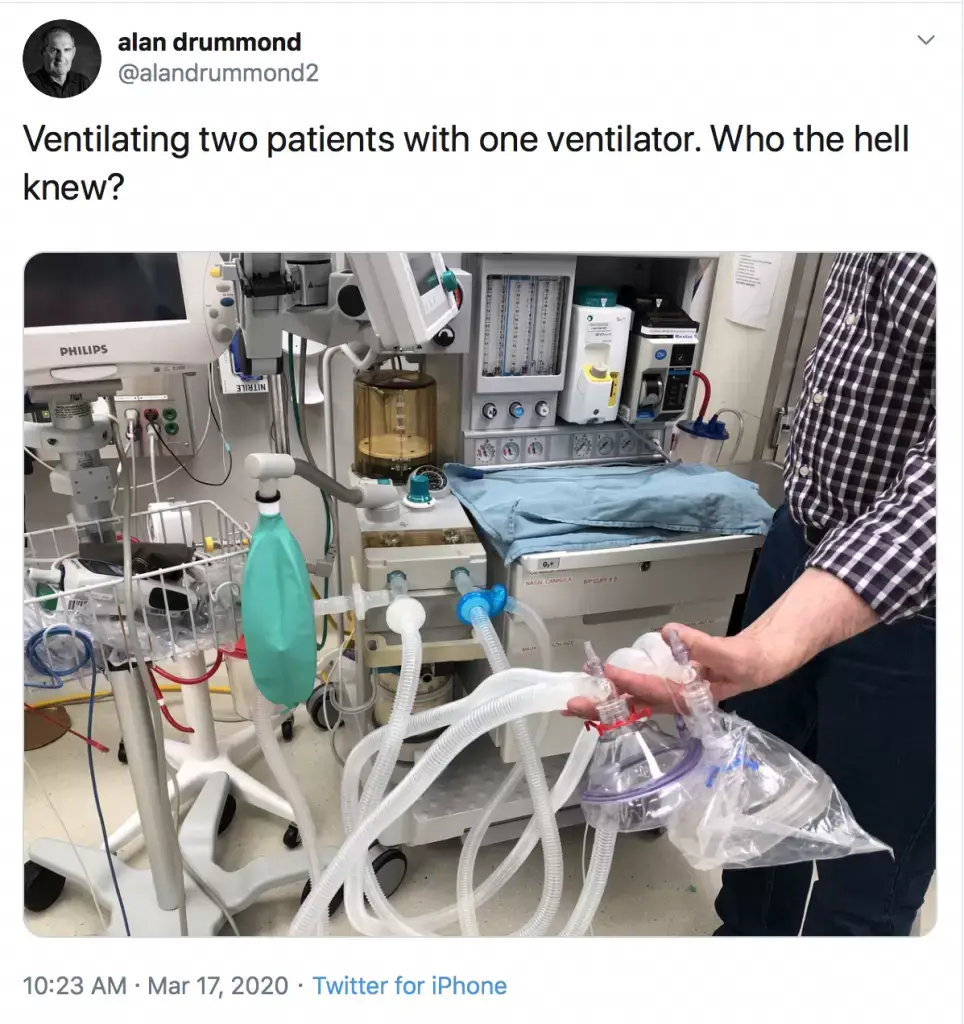
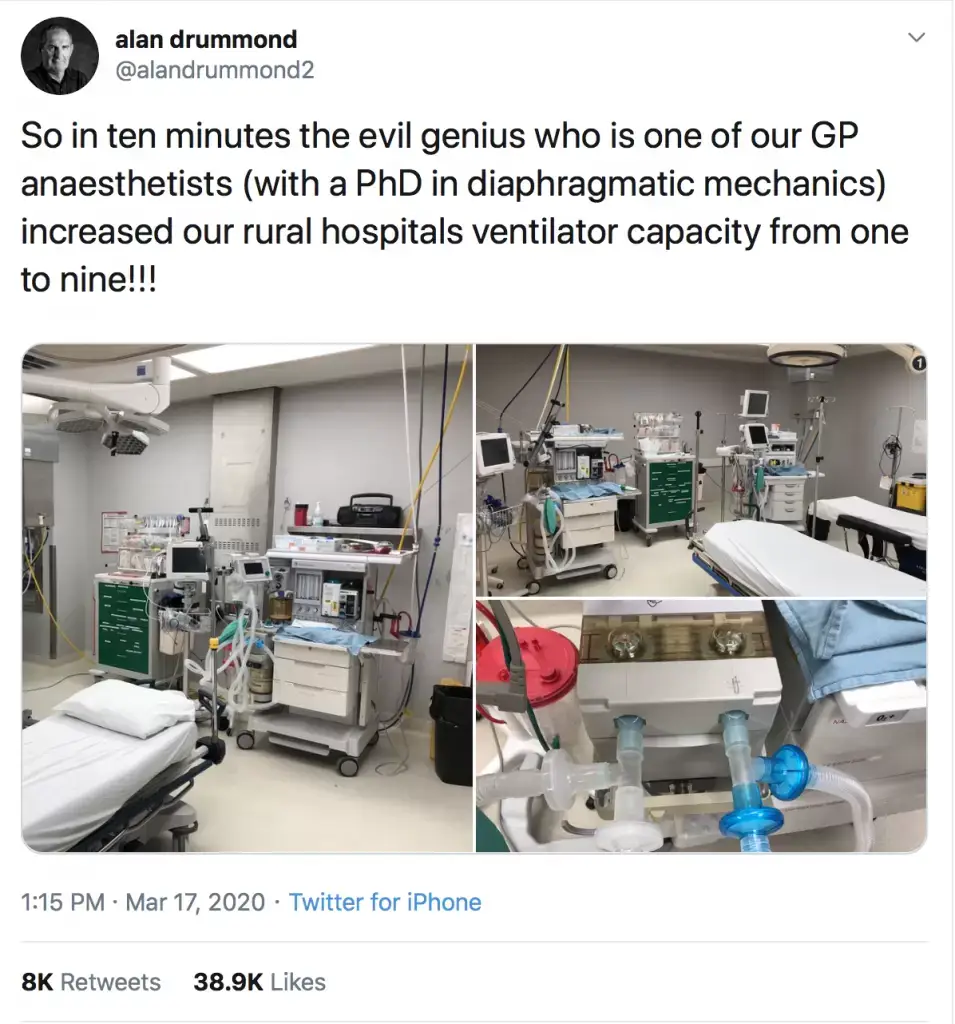
- March 17, 2020: Rural physician Alan Drummond is doing this in Ontario, Canada.
- Peter Attia MD reports an ICU at a hospital in New York has also started “double venting,” putting two patients on a single ventilator.
Practical Problems Still to be Solved
- Cross-Contamination — In running this concept by various individuals, the concern about cross-contamination keeps coming up. One executive at a major healthcare company suggested that the problem could be easily solved with a one-way value made of anti-microbial materials in combination with an anti-microbial air filter on the ventilator’s air intake port. I gather such a filter may not be a standard feature.Update 3/20/2020 — It turns out that one-way respiratory valves already exist and are available here. These look very similar to the one-way valves on first responders’ CPR masks and bag valve masks. I’m unclear if these are sized for or designed for the pressures used in intubation ventilation. I’m hoping someone with more knowledge than I can help me answer that question.REQUEST #1: Someone should adapt a pre-existing HEPA or antivirus air filter to cover the air intake port of a ventilator.
- Ventilation Tidal Volume Variance between Patients is too Great — In a research paper, Richard D Branson et al. did a repeat of Neyman and Babcock’s simulation and published the ventilation performance for each simulated patient: Use of a single ventilator to support 4 patients: laboratory evaluation of a limited conceptTheir conclusion was that the variance of tidal volume (how much air each patient receives per breath) was too varied. They proposed an avenue worth further exploration that would resolve this problem. In their view, it would make sense to have each patient on a separate or semi-separate circuit. They reference that all patients have two lungs (right and left side). In some patients, with asymmetrical lung damage, it’s important to ventilate each lung with different settings. Apparently, this has been done with a single ventilator that splits the pressure produced by the ventilator, but each lung is regulated by a separate control mechanism. They propose that, if such a control mechanism could be adapted, it would eliminate the disadvantages of a simple two- or four-way split.The relevant publications describe using a single ventilator to ventilate the left and right lungs separately with different settings (in particular, PEEP). This same mechanism could, in theory, be used to ventilate two or more patients from a single ventilator with a higher degree of independence.
- Powner DJ et al. Differential lung ventilation with PEEP in the treatment of unilateral pneumonia
- Cavanilles JM et al. A selective ventilation distribution circuit (S.V.D.C)
- Gallagher TJ et al. A simplified method of independent lung ventilation
REQUEST #2: Someone with a mechanical or electrical engineering background should look into the independent circuits used to ventilate the left and right lungs separately with a single ventilator as demonstrated in prior research (the links above) and find a way to adapt it to allow a single ventilator to vent multiple patients with some degree of setting independence for each patient.
Josh Farkas from PulmCrit asserts in his article Splitting ventilators to provide titrated support to a large group of patients that such variances in tidal volumes are not ideal but, if done correctly, tolerable. Specifically, he argues that a mild degree of hypoxia (insufficient oxygen) or hypercapnia (too much carbon dioxide in the blood) would be tolerable in a mass-casualty incident.
Update March 20, 2020
In circulating these ideas with various respiratory therapists, professionals, and ventilator manufacturers, others have identified another way to increase intubation (tube down your throat) ventilation. The idea is to use non-invasive ventilators, which are used with a face mask, and hook them up to an endotracheal intubation tube instead. I’m still in the process of compiling the research on this use case.
Researchers Jagdish Rawat et al. did a successful pilot test of this approach: Role of BiPAP applied through endotracheal tube in unconscious patients suffering from acute exacerbation of COPD: a pilot study
I’ve run this by a respiratory therapist and a few doctors. So far, the thought is that this approach could work in a pinch, but it isn’t a conventional configuration and it’s unclear how many of these devices are in hospitals. You would specifically need a BiPAP ST — a device that allows the machine breaths to be set to a timer (that’s the “T” in “ST”) as opposed to mirroring the patient’s natural, spontaneous (that’s the “S” in “ST”) breathing pattern.
Given all the discussion of splitting a ventilator, several medical association groups have developed consensus statement on this approach. In short, they think it is a bad idea and it is better to give a single ventilator to the patient most likely to survive and no ventilator to the patients that might otherwise share the ventilator.
Here’s the consensus statement against sharing a ventilator from The Society of Critical Care Medicine (SCCM), American Association for Respiratory Care (AARC), American Society of Anesthesiologists (ASA), Anesthesia Patient Safety Foundation (ASPF), American Association of Critical-Care Nurses (AACN), and American College of Chest Physicians (CHEST).
All I can say is I would hate to be the ICU doctor that has to choose who lives and who dies.
Acknowledgments
Thanks to Jaimie Fulton, Respiratory Therapist, Alex Lickerman MD, and Franz Wiesbauer MD & CEO of Medmastery for helping in thinking through this problem, pointing out specific tradeoffs and areas of concern which have been reflected in revisions to this article.
[Medmastery is a medical professional continuation company that is granting medical providers treating COVID-19 patients free access to cross-training for medical professionals on how to operate a ventilator and work in an ICU-like environment. Department heads can request a fee waiver for up to 20 people in their department here.]
About Me
By day, I work as a CEO coach in the Software as a Service (SaaS) industry. I work with fast-growing technology startups to create and operationally handle surges in customer demand. I’m the author of Extreme Revenue Growth and The Recession-Proof Business.
By “night,” I volunteer as an emergency worker in Kitsap County, WA. I’ve trained in mass-casualty incidents, basic life support emergency medical response, and search & rescue.
Additional Resources
If you enjoyed this article, I recommend joining my email newsletter. You’ll be notified when I publish other articles and helpful guides for improving your SaaS business. Submit the form below to sign up. Also, use the email icon below to share this article with someone else who might find it useful.
If you’re the founder and CEO of a SaaS company looking for help in developing a distribution channel strategy, please Click Here for more info.
How to Scale and Grow a SaaS Business


Copied from my comment on Reddit:
Probably late to the party here, but as an RT I figured I’d offer this. It started as a response to a similar thread in a Facebook group of military RTs, but I’ll try to make sure it is accessible for the common reader.
Anyone who has been deployed should have gone through these scenarios in theory during MASCAL (mass casualty) train up.
One of the biggest problems is that these scenarios with test lungs don’t show variability in lung dynamics.
It’s one thing to say you could do this for patients of roughly the same body size / lung volumes in a battlefield scenario. The patients are generally young-ish with generally healthy lungs (barring the occasional inhalation injury, etc).
Lung dynamics of an ARDS patient (e.g. COVID-19) are vastly different. Compliance will vary between different lung fields, between each lung, and between each patient.
In a shared circuit setup, with such variability, unequal delivery of tidal volume is almost guaranteed. (The areas of each lung, per patient, that are not consolidated from fluids/pneumonia/etc, will become hyperexpanded and the areas of the lung that are worse will be hypoventilated.)
One of my biggest concerns is people comparing the use of pressure control vs volume control.
(This is the means by which the ventilator controls each breath. In volume control, a set volume is delivered with each breath. In pressure control, an inspiratory pressure is set for each breath. For each mode, the opposite becomes variable. So in volume control, the ventilator will adjust the pressure up or down to achieve that volume. In pressure control, the tidal volume delivered will vary with each breath.)
Pressure control is the only mode I would recommend, depending on the capabilities of the vent you’re using.
Here’s a hypothetical for you: suppose you’re ventilating 2 patients. you’re in volume control, set at 1000ml for a presumptive 500ml per patient. What if one of those ET Tubes becomes linked? Or occluded? Now that entire 1000ml gets delivered to one patient. A patient who may only have one functional lung. That’s a recipe for disaster.
This setup is a triage setup. It’s meant to stabilize and prepare for transfer to a higher echelon (level) of care.
Management of these patients in an ICU would be a nightmare. Patient-vent synchrony is impossible. One vent change would affect each patient, so titration of blood gases would be nigh impossible.
I think these are good discussions to have, but as a long term solution for a national vent shortage this is a poor solution.
Utilization of this technique should be limited to short term triage, and identification of quick turn around to 1:1 patient:vent ratio, either through transfer of the patient to an alternate treatment facility or acquisition of additional equipment.
Proper management of multiple patients with a single ventilator would require a redesign of the ventilator itself, and development of a new machine capable of monitoring and controlling multiple circuits at once.
Nex,
Thanks for contributing your experienced perspective. As a result of these conversations popping up everywhere, I understand engineers at multiple ventilator companies are looking at designing a more elegant way to vent multiple patients from one vent. I agree the arterial blood gas management would be a total pain on a extended care basis.
‑Victor
Pingback: How to Increase Ventilator Capacity 2–4x in 10 Minutes – The Blog of Author Tim Ferriss | A wealthy life
Found this 3D printing CAD model of a respirator valve and mask if anybody finds it useful…
https://grabcad.com/library/respirator-valve-and-mask‑1?fbclid=IwAR01pBrLIE2G2o8N0XK9nXW529_kUfUPs3EF0iBXpuhO7efRiTUObiakShk
Hey Victor,
This is great, I will make sure to share as much as possible.
One thing I was thinking is if the problem of matching up size and lung compliance could be solved using inexpensive PVC Ball Valves or something of the sort found in emergency rooms to limit the flow of air going into the tubing. Another option is that by extending the length of the tubing the airflow volume might be less given that it would be traveling a longer distance hence adapting to the size and lung compliance?
I know this can’t be known for sure without testing but this testing could be done using their same methodology to check the volume reaching each lung. If this works reliably it could save even more lives.
I might try and get in contact with a friend in health care to try to test this idea to get a reliable amount of oxygen that could be measured to match the need of patients.
Keep doing the awesome work. ?
Victor,
We are trying to build & donate 1,000s of 4‑way splitters so that people don‘t die due to shortage of Ventilators. It would be highly appreciated if I can speak to you or the Doctors who have done successful splitting in the past. Plz check this thread:
https://twitter.com/pg211183/status/1241574015933779968
Hi Victor,
Please disregard my last comment. I read your article a day after it first came out and did not realize it was updated to address the contamination issue. I have seen the update and wish to take back my last message.
Hi Victor,
This idea was evaluated by anesthesiologists and microbiologists and has been found to be *unworkable due to the risk of contamination between patients.*
As this is a highly sensitive topic, please consider more medical professional consultation and update your article based on those results.
I applaud your effort to help with the situation and agree that this is a very interesting idea but unfortunately it is more complicated than it seems from this analysis.
Big fan of your work!
To get ‘independent circuits’ the cheapest and quickest way would be by adding or removing a constriction, for example: using a valve; squeezing a hose; using a longer hose; using a shorter hose; a bigger diameter hose would give different flows in each hose
— the tricky part is measuring and managing the flow through each hose; if you only need to be 60% right then this should be ok but if you need to be 95% right then it’s tougher
— leaving one hose free to let out excess air would make the other hoses easier to control
Pingback: Emergency Technique: How to Increase Ventilator Capacity 2–4x in 10 Minutes – The Great Self Improvement
Pingback: Emergency Technique: How to Increase Ventilator Capacity 2-4x in 10 Minutes – The Blog of Author Tim Ferriss
Pingback: Open Source Respirator and Low Cost Ventilator Efforts to Fight Coronavirus (COVID-19) | Innovation, Change and Digital Transformation
Very useful and well written article!
One small recommended correction:
in the line: ” 285 kg x 2.2 kg per lb = 627 lbs combined patient ideal body weight,”
the unit conversion should be 2.2 “lb per kg” instead of “kp per lb” (a kilogram is heavier than a pound.)
Joseph — Thanks for catching the typo. I revised it.
‑Victor
We thank you for your heart towards all people on this earth!
You truly are walking in purpose!!
🙂
Great article, Victor! These ideas need to be spread in the mainstream during this critical time, because this is REAL NEWS.
Placing appropriate check valves on each arm of the augmented ventilator might inexpensively reduce cross-flow and mitigate cross-contamination among patients sharing a device. While the wikipedia entry for this device looks complex, simple biological versions exist in each human heart. Medically, such valves are not uncommon.
https://www.harvardapparatus.com/one-way-respiratory-valves.html
https://www.ncbi.nlm.nih.gov/pubmed/20810677
Brad — Thanks! This is very helpful. I revised the post to include this info. It looks quite similar to the one valve on my CPR mask. ‑Victor